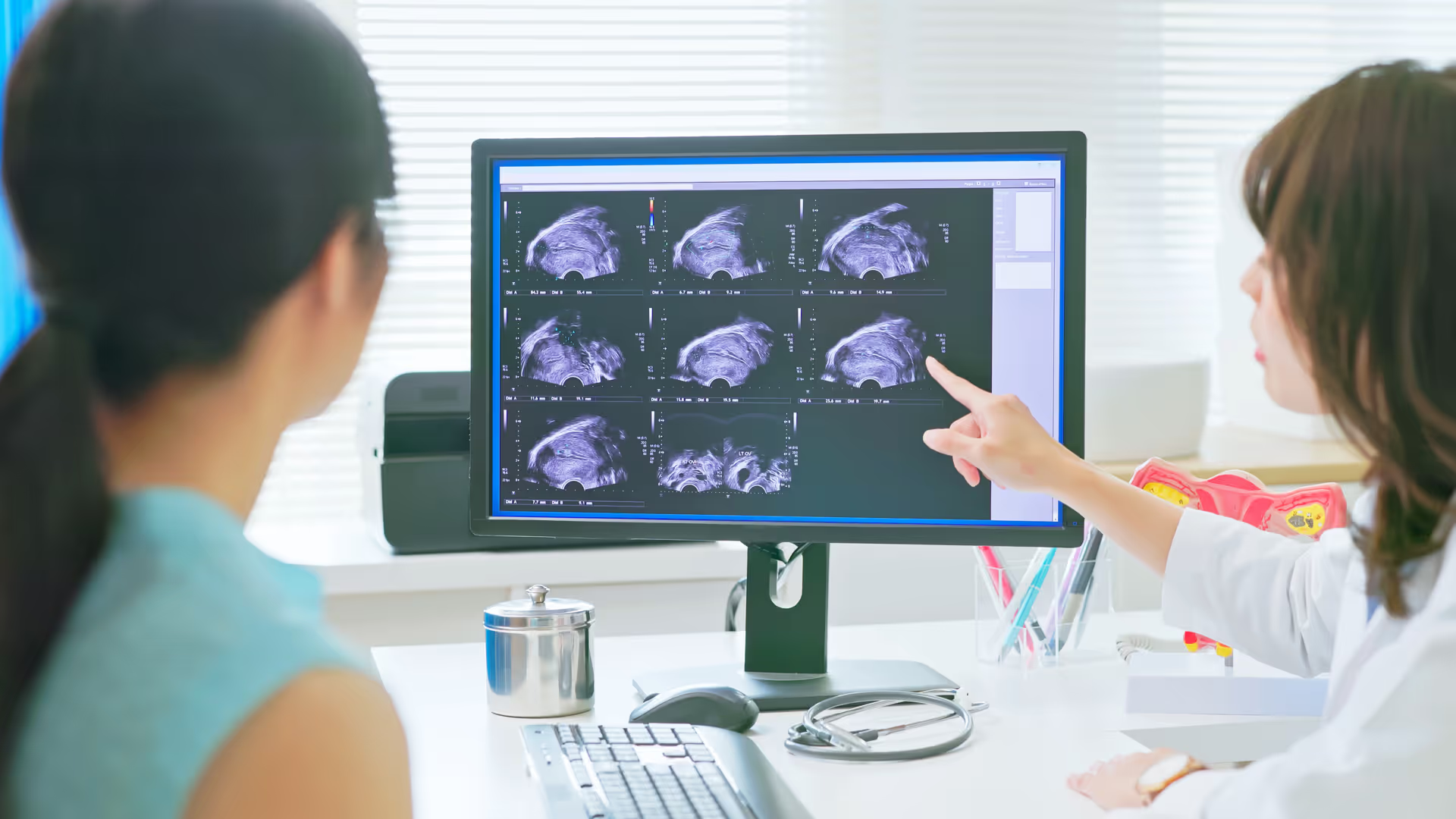
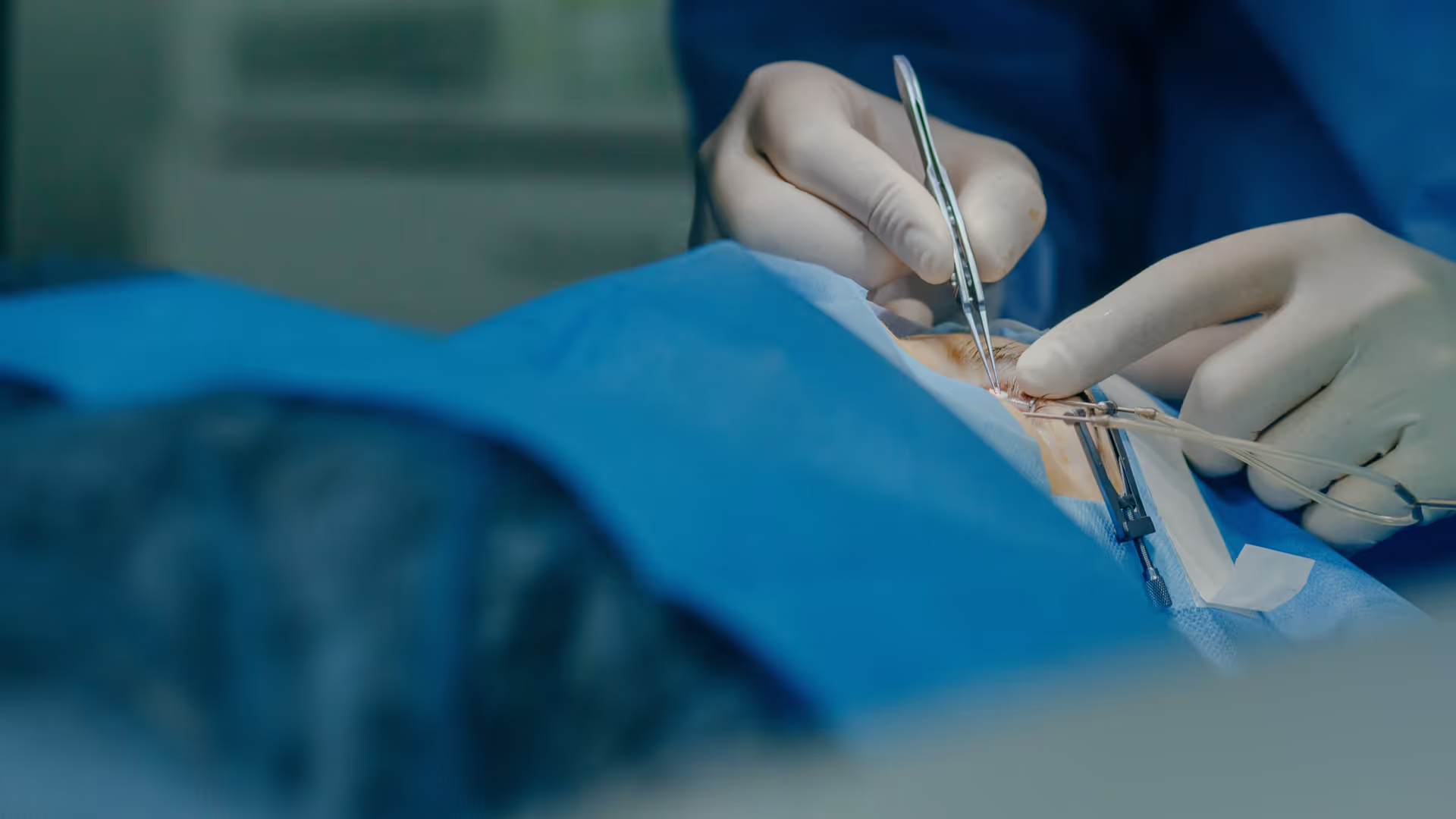
Insights and resources
Thank you! Your submission has been received!
Oops! Something went wrong while submitting the form.
Load more
1 / 36
Talk to us about your next project
We help clients with all stages of their most complex and challenging technology and product development projects.
If you're considering the next steps along your innovation journey, why not get in touch?
Get in touch
Talk to us about your next project
Whether you would like to discuss a project or would like to learn more about our work, get in touch through the form below.