
Medical device manufacturing
Bridging design, prototyping and manufacture – from clinical builds to scale-up
The hardest part of manufacturing isn’t volume, but the transition from design into regulated production. TTP bridges that gap by combining medical device prototyping with manufacturing process development, supporting agile clinical and early production builds. With no minimum volumes, no CMO lock-in, and no process IP constraints, you retain control of your product and supply chain. We help you reach the clinic sooner, and help you transfer to full-scale manufacturing with confidence, without late-stage surprises.
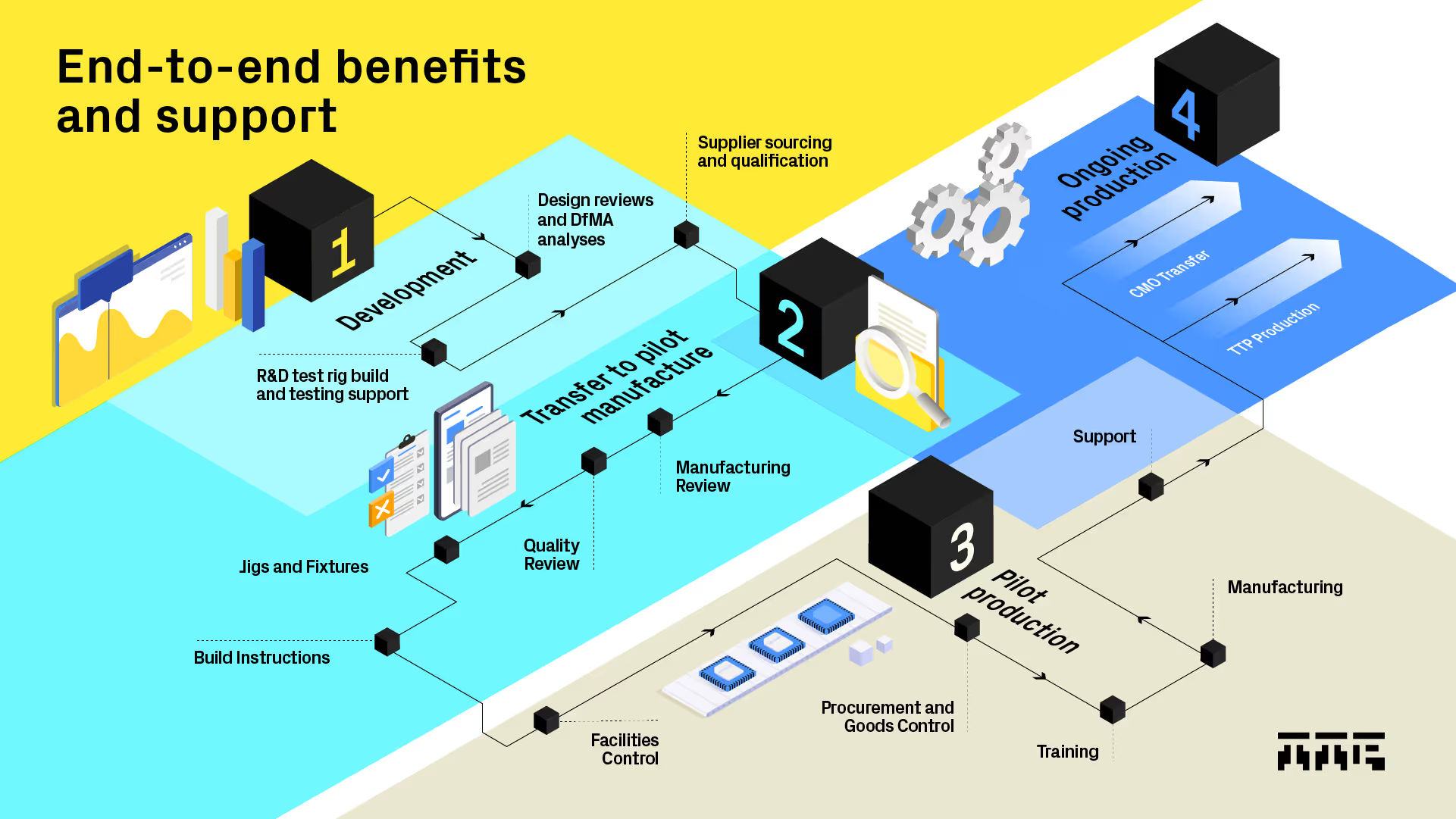

Design and manufacturing, tightly integrated
When design and manufacturing work as one team from the start, you avoid the disconnects that often create delays, rework, and transfer headaches later on.
TTP supports you with:
- Manufacturing input built into design reviews from day one
- DFM/ DFA insight to make designs production-ready early
- Faster resolution of build issues through close collaboration
- Reduced risk at verification and design transfer
- One continuous journey from prototype to clinical builds
Because we are not a high-volume CMO, our incentives are aligned with yours: to design a process that will scale cleanly when the time is right, not to pull you prematurely into fixed production routes.

The team at TTP has done a fantastic job expediting the journey to commercialisation of our prototype. We are excited about the potential for Seracam to improve patients’ lives."
Dr Paul Cload
Chief Marketing Officer
,
Serac Imaging Systems
TTP really bridges the gap between true product development and true manufacturing. Their team understands the science, the design trade-offs, and what it takes to bring a novel device into production.
Paul Goode
CEO
,
Glucotrack

TTP has been an incredible partner, complementing our expertise and delivering designs from whiteboard to moulded functional prototypes. With TTP’s support, Achilles Therapeutics is scaling its manufacturing process and bringing transformative therapies from bench to bedside. The TTP team has shown the same level of excitement as us in getting these therapies to the people who need them.
Ed Samuel
EVP Technical Operations
,
Achilles Therapeutics


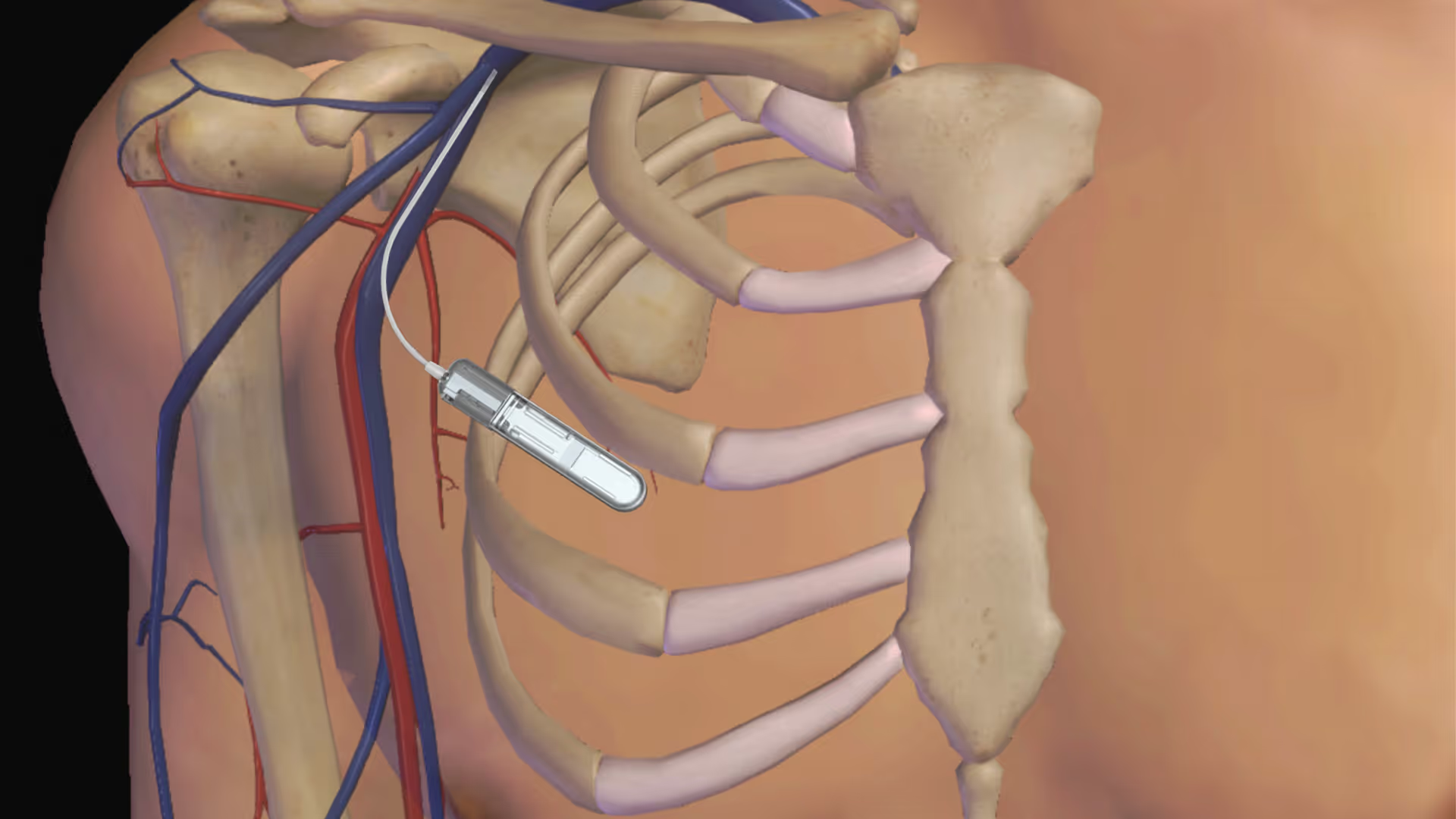
Rapid builds for clinical momentum
As you approach verification or clinical studies, speed isn’t about rushing, but avoiding months lost to external delays, or slow disconnected teams. Cleanroom and microfabrication capability onsite enables tighter process control for devices incorporating microfluidics, sensors, MEMS-like structures, or precision assemblies, where early manufacturing conditions materially influence performance.
TTP helps you maintain momentum with:
- Short lead times for verification and clinical builds
- Dedicated manufacturing facilities spanning 1,100m², including cleanrooms, microfabrication, assembly, and other specialist labs
- Faster build–test–refine cycles under one roof
- Rapid response to engineering changes when designs evolve
- Controlled builds that generate trusted clinical evidence
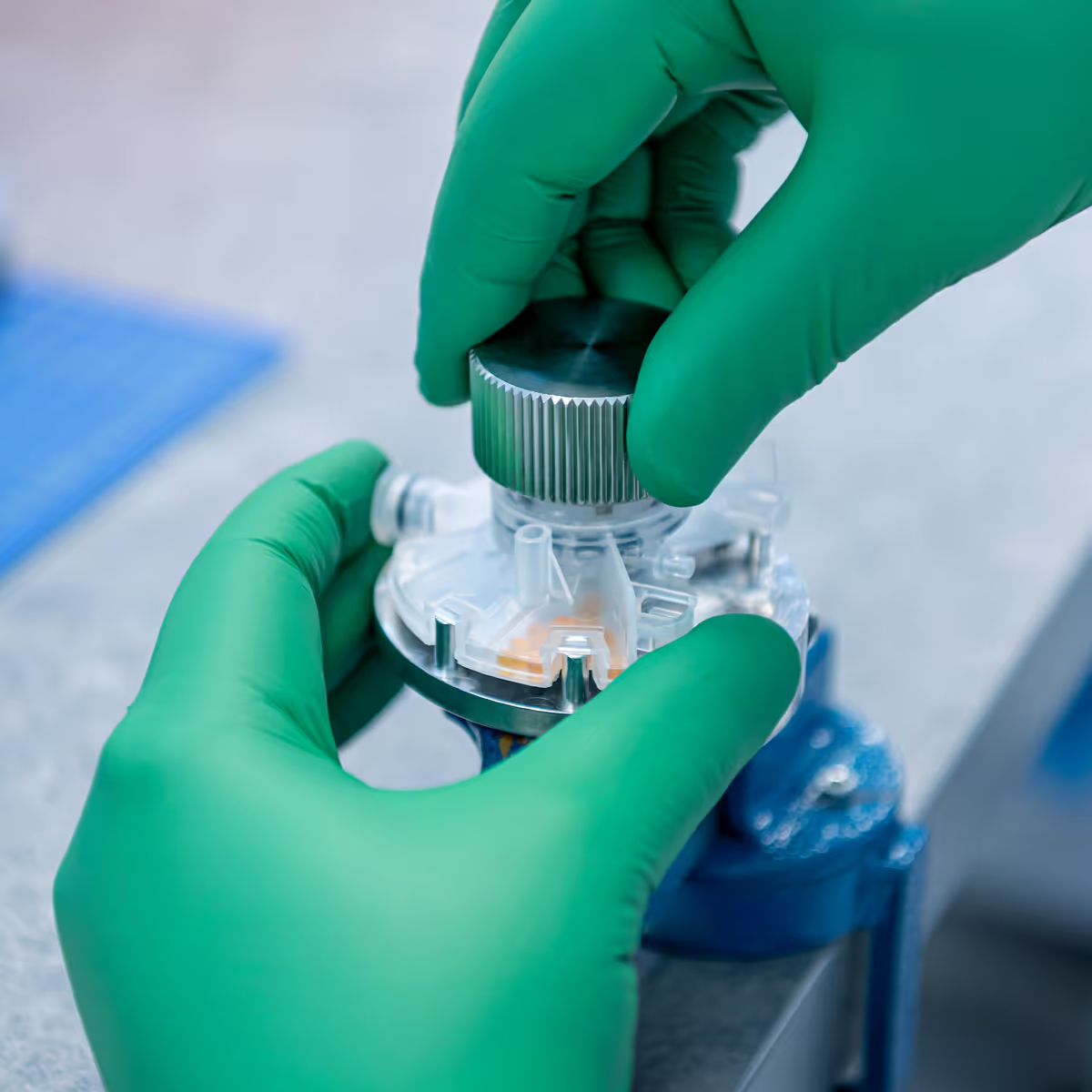

No minimum volumes, no locked-in commitments
Early-stage devices rarely follow predictable demand curves. Manufacturing should flex with your project, not force you into thresholds or long-term commitments.
With TTP, you can:
- Build single units or early series volumes with no minimum order
- Scale from prototypes to ~100,000 low-complexity devices annually, and hundreds to low thousands for high-complexity systems
- Support uncertain adoption curves, novel products, or emerging markets
- Retain full ownership of product and process IP
- Delay final CMO selection until design maturity, enabling a true competitive bidding process
This gives you strategic freedom: validate the design, mature the process, and then select the right long-term manufacturing partner based on data, not dependency.


Scale-up when you’re ready
The goal isn’t to transfer to a CMO quickly; it’s to transfer at the right moment, with the processes, evidence, and confidence needed to scale smoothly.
We can help you with:
- Manufacturing process development and validation alongside the product
- Supplier identification and selection informed by hands-on build experience
- Documentation and data packs to support design transfer
- Training and collaborative builds with your chosen CMO
- A smoother pathway from clinical supply to high-volume manufacture
We have successfully transferred programmes to external CMOs following clinical and early commercial builds, with validated processes, qualified suppliers, and clean handovers, reducing the typical friction seen when DfM is theoretical rather than proven in practice.


Regulatory-grade manufacturing confidence
For MedTech, manufacturing isn’t just about making units; it’s about building in a way that stands up to scrutiny. We provide:
- ISO13485-certified manufacturing where required
- Full traceability, batch records, and controlled processes
- Cleanroom environments aligned with medical device standards
- Evidence that supports verification and clinical builds
- Access to trusted partners for sterilisation and low-volume fill–finish where needed
How we can help
Clinical builds
Small-batch manufacture to support verification and clinical studies, delivering controlled builds and traceable data suitable for regulatory scrutiny.

New process development and validation
Development and optimisation of manufacturing processes alongside device design, with validation to ensure repeatable, scalable production.

Design integration
Close collaboration between design and manufacturing teams to resolve build challenges early and ensure designs are ready for efficient production.

DFM/ DFA
Design for Manufacture and Assembly insight embedded from the start, reducing complexity, improving build efficiency, and supporting smooth scale-up.

ISO 13485 compliance
Manufacturing within an ISO13485-certified quality system, ensuring full traceability, controlled processes, and compliance with medical device standards.
Read more about our approach to regulatory compliance

Complex instrument and device assembly
Assembly of sophisticated electromechanical devices and medical systems requiring precision, specialist processes, and multidisciplinary expertise.

Filling and sterilisation with our partners
Access to trusted partners for sterile fill–finish and sterilisation services, integrated into the manufacturing pathway where required.

Microfabrication
Our in-house microfabrication facility enables fast development of micro-devices, alongside a global network of foundries to allow production transfer of the devices we develop. With processes across silicon, glass and polymers we offer a unique environment to apply the benefits that advanced microfabrication offers in sensors, microfluidics and surface modification technologies.

Manufacturing transfer readiness
Process documentation, supplier selection, and data packages prepared to enable efficient transfer to your chosen high-volume manufacturing partner.

Clinical builds
Small-batch manufacture to support verification and clinical studies, delivering controlled builds and traceable data suitable for regulatory scrutiny.

New process development and validation
Development and optimisation of manufacturing processes alongside device design, with validation to ensure repeatable, scalable production.

Design integration
Close collaboration between design and manufacturing teams to resolve build challenges early and ensure designs are ready for efficient production.

DFM/ DFA
Design for Manufacture and Assembly insight embedded from the start, reducing complexity, improving build efficiency, and supporting smooth scale-up.

ISO 13485 compliance
Manufacturing within an ISO13485-certified quality system, ensuring full traceability, controlled processes, and compliance with medical device standards.
Read more about our approach to regulatory compliance

Complex instrument and device assembly
Assembly of sophisticated electromechanical devices and medical systems requiring precision, specialist processes, and multidisciplinary expertise.

Filling and sterilisation with our partners
Access to trusted partners for sterile fill–finish and sterilisation services, integrated into the manufacturing pathway where required.

Microfabrication
Our in-house microfabrication facility enables fast development of micro-devices, alongside a global network of foundries to allow production transfer of the devices we develop. With processes across silicon, glass and polymers we offer a unique environment to apply the benefits that advanced microfabrication offers in sensors, microfluidics and surface modification technologies.

Manufacturing transfer readiness
Process documentation, supplier selection, and data packages prepared to enable efficient transfer to your chosen high-volume manufacturing partner.

Speak to one of our experts

Will Cramer
Will Cramer leads TTP Manufacturing, delivering low to medium volume production of consumables, devices, and instruments to ISO13485 standards. A mechanical engineer with over 25 years’ experience across medical, life science, consumer, and industrial markets, he previously served as VP Engineering and Production at a TTP spin-out. Will holds a master’s in engineering from Cambridge University.


Fred Hussain
Fred leads TTP’s microfabrication capability as part of TTP Manufacturing.He has 30+ years’ experience taking devices from lab toscale-up.Hehas delivered 100+ internal/external projects, mainly in the bio-med sector.Fred has a Master of Engineering degree in Electronics and a PhD on 3-axissilicon accelerometers.


Sophie Meredith
Sophie is an experienced project leader in TTP’s Biosensing team. She has a PhD in biophysics and has worked on projects involving the development of optical systems, implantable devices, and fluorescence detection for applications in biological sensing


Justin Buckland
Justin is the technical lead for multiphysics analysis and sensor development at TTP. He holds a PhD in Surface Physics from the University of Cambridge and has played a pivotal role in the development of a wide range of sensing technologies including microfluidic, electrochemical, acoustic and piezoelectric.

Meet some of the team

Will Cramer

Fred Hussain

Sophie Meredith

Justin Buckland
The medical device development process
Medical device development is a structured yet flexible journey - one that demands meticulous planning, a clear understanding of risks and trade-offs, and a focus on quality and regulatory requirements and compliance. Here are our five phases of designing and developing a medical device at TTP.

Human-centred design and human factors engineering
At TTP, we design technology around its users because usability is essential, not optional. Our Human Factors team combines behavioural science, engineering and design to create solutions that are safe and widely adopted. Through human centred design and engineering, we help clients reduce risk, move faster and deliver market ready devices.


















