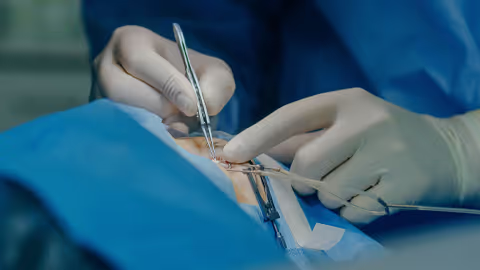
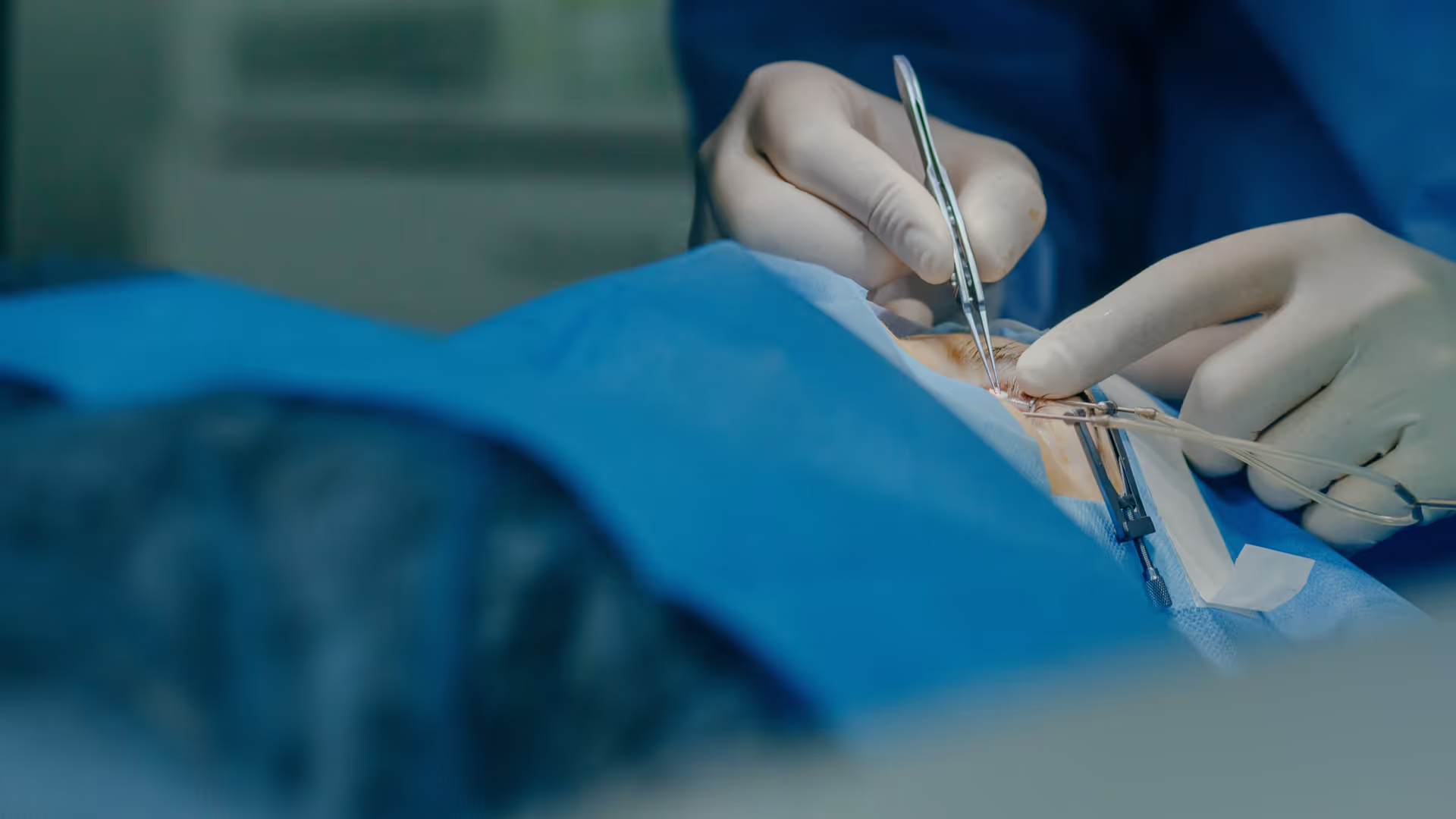
There’s a lot of excitement at the moment about sustained-release of drugs to the back of the eye. In addition to established names such as Ozurdex and Iluvien, companies like Ripple Therapeutics, Re-Vana, EyePoint – and many others – are developing new approaches to increase drug loading and improve control over release rates. Even better, many of these therapies are designed to fit within established in-office procedures to make adoption easy.
This is a good news story. However, many of these companies have found that the “easy step” of creating a robust and reliable inserter device to deliver an implant into the vitreous is harder than it first appears. TTP has worked on five different solid-dose intravitreal applicators – from ground-up development through technical review to in-production troubleshooting – building deep expertise in inserter design along the way.
This insight piece shares some of that expertise: if you want a smoother path to a compelling solid-dose IVT inserter, here are four things worth considering.
The anatomy of an IVT inserter
But before exploring the key design considerations, it may be helpful to look briefly at the anatomy of IVT inserters.
An IVT inserter is – in principle, at least – not a complicated thing: it is a needle containing an implant, and a push rod with which to eject it. But, as is often the case, the devil is in the detail.
Beyond the basics, an effective inserter must achieve several other things.
- It must keep the implant in place until the user actuates the device
Anyone who has handled implants will attest that they sometimes seem to have a life of their own! They will sometimes stick to things (they are often quite susceptible to static)… and then fall off for no apparent reason. The same is true in a device – the implant might seem perfectly happy where it is, but a fair proportion of them will just fall out when you pick the device up.
- The implant ejection speed must be controllable
The lower limit to ejection speed is set by the need for the implant’s momentum to carry it clear of the needle and push rod to avoid “track-back”, the phenomenon in which the implant follows the needle track out of the eye when the needle is withdrawn. The upper limit is defined by the need to avoid the implant travelling right across the vitreous and hitting the retina. These limits depend on the specific diameter and length of the implant itself. For an IVT, in contrast with introducing an implant into the anterior chamber, these limits are quite well spaced.
- The device must be economical to produce
While this does have implications for the entire design, the most important factors are that the insertion of the small, fragile implant into the device should be achievable by automated equipment, and that the design supports primary assembly on one site and implant installation in another (if required). As always with such things, device design decisions can make this either much easier – or much more difficult.
1. Implant retention
If you’ve not worked on solid-dose inserters before, the idea of needing a mechanism just to keep the dose inside the device might seem odd; there’s no real equivalent with liquids. In fact, early development of implanters is often dominated by the opposite question: how do you get everything to line up so that the dose actually comes out?
But, jamming and static notwithstanding, retaining the dose reliably in storage and during the early stages of use – and then releasing it smoothly when required – is a surprisingly significant challenge.
In a sense, this breaks down into two possible requirements: retention of the implant during transport, storage, and prep; and retention immediately prior to injection, once all prep steps – removing a cap, for instance – have been completed.
While both are obviously desirable, the first is both far easier to achieve technically – with a “pin” in the cap blocking the needle exit, for instance – and far harder to achieve by, say, training or user instructions alone. Whether to design for both should therefore be a deliberate choice.
Key design choices
A key design choice is whether to retain the implant within the needle itself, or further back in the device, outside of the needle. Both approaches have pros and cons.
Retention within the needle either requires something reaching in from the outside (the gel plug used in the Durysta IC device, (for example) or implies a custom needle, with all the associated cost, time, and supply chain implications. On the plus side, it pushes the challenge of loading a fragile implant into a very small tube into the automation environment, where every advantage of precision and imaging can be brought to bear.
By contrast, retention outside the needle allows the use of off-the-shelf needles but shifts the alignment challenge into the device itself. That means achieving precision at the tens-of-microns level in the field using plastic parts: far from impossible, but a significant engineering challenge which must be treated as a central design driver.
The last big choice is between force-based retention (such as the Ozurdex-type rubber sleeve pressing on the implant) and displacement-based retention (where the path is simply blocked until actuation).
Force-based systems are typically simpler since they are "always on” : they do not need to be released prior to actuation. On the other hand, that also places an upper limit on the retention force they can provide, and makes them more susceptible to vibration and shock - passing some of that burden on to be partly solved by secondary packaging.
Displacement-based systems generally involve more moving parts, but the risks that need to be proven out for them are typically more concrete and familiar: will this part move by at least this distance? If well designed, they are usually more robust and are typically more accommodating of different-sized implants within a single design, with minimal per-size changes.
2. Easy automated implant insertion
Familiarity, dexterity, and practice can make it easy to overlook just how tiny many sustained-release implants are. They are also often quite fragile – typically either glassy and brittle or rather crumbly. When you consider that these tiny implants need to be aligned with correspondingly tiny needles and retention features, it quickly becomes clear why this is not easy.
This challenge lies at a crossover between device engineering and automation engineering; we’ll focus here on the aspects that have device implications for now, but the automation challenges should also be considered in project planning.
For the moment, let’s say that the design involves installing the implant into the needle. The essential requirement is to achieve positional tolerance perpendicular to the needle of a few tens of microns, and orientation precision of a couple of degrees. Tight, but not unreasonable for specialist equipment – or even using a precision v-groove.
Precision and alignment tolerance
The real difficulty is that the alignment tolerance between the steel tube and the hub in standard needles is nowhere near this precise: in practice, the hub is of very little use as a reference.
This leaves two options: physical alignment on the tube, or optical alignment and a well-registered precision actuator.
Physical alignment coupled with “muzzle loading” (inserting the implant through the sharp end of the needle) is technically not complicated– a single precision-made block enables each cycle to be quite “dumb” – but it does present questions around potential damage to either the needle tip or the implant, which need to be addressed in the detailed process design.
“Breech loading”, by contrast, avoids those risks, but requires a more complicated precision block to hold the needle on one side of the hub while inserting the implant from the other side. It also requires care to avoid bending the needle during the operation.
Optical alignment is simple in principle, though in practice drift means frequent re-calibration will be required, alongside the cost and overhead that more complex automation stations inevitably bring.
Turning attention briefly to designs which involve installing the implant outside of the needle, the precision requirements simply transfer to the registration between the component holding the implant and the needle itself: a breech loading approach still requires considerable thought and care, but is achievable.

3. Design for two-site assembly
As with a lot of drug delivery devices, it’s often advantageous – or even necessary – for most device manufacture and assembly steps to take place at one facility, with installation of the drug happening elsewhere. This means that the device, as shipped, needs to be coherent and robust, with all device CQAs determinable at the primary device assembly site – even more so if the device manufacture is outsourced.
The best approach to this depends significantly on how the implant is retained and installed. A force-based retention coupled with a muzzle-loading installation enables the device to be shipped from primary assembly intact, apart from a cap. A displacement-based retention with the implant stored outside the needle could also be shipped essentially complete if a suitable shuttle component were used, though this relies on that feature being designed in from the beginning of development.
Retention, ejection and interlocking considerations
Whatever the details of the design, several things must be achieved. For one, both the retention and the ejection mechanisms must be testable, with confidence that their functionality will not change during final assembly – or the design and both stages of assembly must be so robustly verified and qualified that there is no need for additional testing.
This consideration can usefully act as a selection criterion when developing options for these mechanisms from the very beginning. For example, the level of testing needed to build confidence that a retention mechanism based on interference with an elastomer would behave identically during both test and final loading would be significant.
Another thing which must be addressed is interlocking. For example, it is quite common to use a cap or a pull-pin to lock the mechanism out during distribution and storage: this is usually no less necessary in shipping prior to final assembly.
Ideally, these interlocks could be engaged during primary assembly. If that cannot be done, transport clips, which duplicate the relevant features can mitigate the risk. However, it is important to think through what happens if the partly-assembled device actuates on removal of the clip.
As is often the case, this is partly about selecting concepts which fit the specific device context, and partly about ensuring that requirements of this kind remain visible and continue to be worked to throughout detailed design.
4. Controlled implant ejection
The final area we’ll consider in this insight piece is the actual ejection of the implant. Current devices use a push wire slightly smaller than the inner diameter of the needle to interface with the implant and push it out– a solid and simple approach. The challenge arises in how this wire moves, and how it is coupled to the control operated by the clinician. How can a device avoid both “track-back” and “overshoot”?
Automatic vs proportional systems
Essentially, the choices are either an “automatic” system (for example, releasing a compressed spring) or a “proportional” system in which the ejection speed is directly controllable by the clinician.
Automatic systems are not commonly considered for the vitreous since the target speed window is wider than for the front of the eye, but they remain a valid option. The primary challenge is that the target ejection speeds are not especially high, while the required travel is still in the high single-digit millimetres (at least), so a combination of variable mechanism friction and the impact of orientation can introduce meaningful variation – though both can be designed around, with care.
Proportional systems are more common and skilled clinicians can use them well, so long as the design achieves a few things. First, the user action must be easy to perform within the speed range which the mechanism requires. Second, the resistance that the mechanism provides must be consistent and avoid sudden drops. Third, the mechanism must be low-friction and sufficiently stiff that it cannot store significant energy by bending – otherwise there is a risk of sudden release, with the implant being ejected far faster than intended.
A final point
If the retention mechanism is of a displacement type then the timing of releasing the retention and actuation must always be right. In practice, this drives design towards a chain of mechanisms actuating each other and away from parallel mechanisms actuated by a single trigger: attempting to eject an implant which is still solidly retained will simply destroy the implant.
In conclusion
Notwithstanding everything already said, it remains true that IVT inserters are not especially complicated devices, and a wide variety of approaches can produce successful designs. The key point of this piece is to highlight some of the difficult-to-foresee issues and so enable designers of new devices to avoid them.
Given the number of companies which have publicly announced that they are working on solid-dose IVTs – to say nothing of those still in stealth mode – there are likely to be several of these new devices emerging in the next few years.
About TTP's Ocular Drug Delivery Team
TTP’s ocular drug delivery team develops advanced front- and back-of-eye delivery systems, spanning topical, intravitreal, subretinal and suprachoroidal therapies. Combining expertise in engineering, fluidics, optics and human-centred design, the team solves complex challenges around precision delivery, usability and manufacturability for next-generation ophthalmic treatments. From early concept development through to scale-up and transfer to manufacture, TTP helps clients accelerate ocular devices and sustained-release systems that improve adherence, clinical performance and real-world outcomes.
Find out more